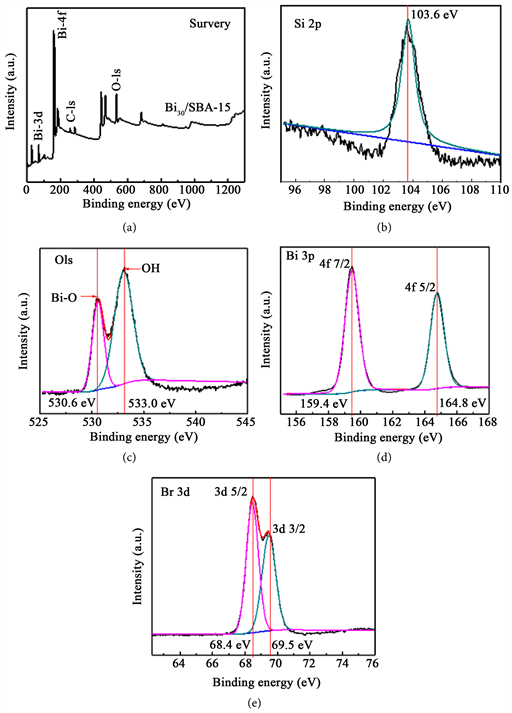
13 Recent development of rare earth and Y–Zr solid solutions and C-type rare earth oxides has marked the activity in the appropriate range for high temperature applications. 12 Among various direct NO catalysts, perovskites exhibit very good activity at temperatures higher than 600 ☌. Catalysts that have been reported to be active in the decomposition of NO can be divided into three major groups: metal oxides, zeolites, and supported metals. 11 Consequently, catalysts that help lower the kinetic barrier is essential to achieve efficient direct NO decomposition. NO is a thermodynamically unstable molecule and its decomposition into elements is favored at temperatures below 1000 ☌, 11 however, the direct decomposition is kinetically hindered due to a high activation barrier of 335 kJ mol −1. 9,10 An alternative solution for NO removal is the direct NO decomposition into harmless N 2 and O 2, which in principle is ideal method for the solution of NO problem. 3 The current techniques used for the abatement of NO such as selective catalytic reduction (SCR) and non-selective catalytic reduction (NSCR) and NO storage-reduction (NSR) all rely on introducing reducing agents such as ammonia, diesel soot, carbon monoxide or hydrocarbons 4–8 to drive the reduction of NO into N 2.

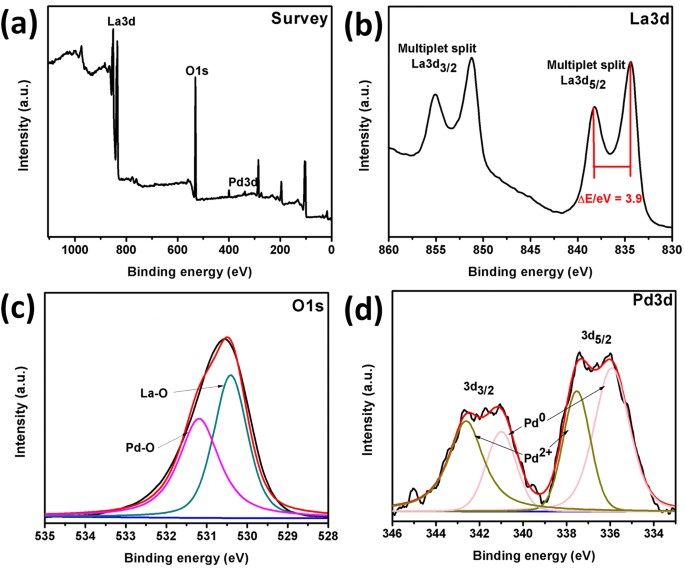
1,2 The laws on NO emissions are increasingly strict and the research of NO abatement remains a hot topic. Among the various pollutant gases, nitrogen oxides are considered as one of the primary pollutants of the atmosphere, since they are responsible for environmental problems like photochemical smog, acid rain, tropospheric ozone, ozone layer depletion and global warming. Introduction The increase of the worldwide population has led to an increase of the energy demand and an increase of environmental pollution. Our results suggest retarding metallic Pd oxidation is key factor to develop a sustainable catalyst for direct NO decomposition at lower temperatures. At temperatures higher than 500 ☌ the PdO converts to metallic Pd by releasing oxygen, resulting in stable direct NO activity. The catalyst pretreated in helium yields Pd–PdO composite structure and higher active metallic surface area compared to the hydrogen pretreated catalyst and exhibited higher NO conversion at lower reaction temperatures (<500 ☌). The oxidation ability of metallic Pd is sensitive to the particle size of the metallic Pd. Combining characterization and catalytic performance, it has been revealed that the reaction in the lower temperature region (<500 ☌), is not catalytic and NO chemically reacts with metallic Pd and forms PdO. Three different types of catalyst compositions of palladium namely Pd, PdO and mixture of Pd–PdO, were created on silica surface by controlling pretreatment environments for systematic comparison. SiO 2 supported palladium was studied as a model catalyst system for direct NO decomposition over temperatures ranging from 100 to 800 ☌ both in dynamic and steady state mode to understand the changes in the palladium chemical state with respect to the reaction temperature. However, the function of palladium during the reaction at various reaction temperatures is still unclear. Supported Pd catalysts have been investigated recently for the reaction ranging from 300 ☌ to 1000 ☌. Although its decomposition is thermodynamically favorable (ΔG = −86.6 kJ mol −1 at 25 ☌), yet kinetically hindered due to a high activation barrier of 335 kJ mol −1, making use of catalyst indispensable. Direct decomposition of NO into N 2 and O 2 offers an ideal solution to the abatement of NO from automotive and various combustion processes.
